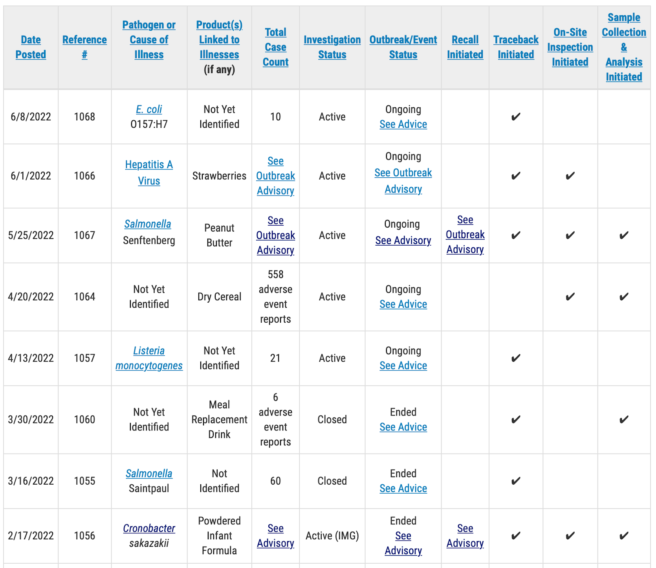
FDA examining brand-new break out of E. coli O157: H7 infections; 10 individuals ill
The Food and Drug Administration is examining a brand-new break out of E. coli O157: H7 infections, however little details has actually been launched.
Ten clients have actually been recognized in the E. coli break out, however no info has actually been launched about where they live or what their ages are.
The FDA reports that it has actually started traceback efforts, however has actually not exposed what food or foods are being traced. In current days the USDA’s Food Safety Inspection Service reported it is examining an E. coli break out, however did not publish any information. The companies have actually not suggested whether the examinations belong.
E. coli is frequently discovered in hamburger, however has actually likewise been discovered in other meats along with in fresh vegetables and fruits. It is prohibited for individuals to offer meat infected with E. coli O157: H7 and numerous other pressures of the pathogen, regardless if they know the contamination.
Hepatitis A break out examination
In another examination, the FDA has actually started a remote Foreign Supplier Verification Program assessment associated to strawberries imported from Mexico. The natural berries are connected to a break out of liver disease A infections in the U.S. and Canada.
Canadian authorities have actually started a different examination.
The natural strawberries were imported to the United States from Baja California, a state in northern Mexico, and branded as FreshKampo and HEB by a typical provider. They were for sale in between March 5, 2022, and April 25, 2022.
However, the FDA is prompting customers who purchased the natural strawberries and froze them for later usage to toss them away. Freezing does not eliminate the liver disease An infection.
As of today the FDA is reporting 17 validated diseases– 15 in California and one each in Minnesota and North Dakota– and 12 hospitalizations in the United States. In Canada, 10 clients have actually been related to 4 of them needing hospitalization.
Listeria break out
An examination into a break out of Listeria monocytogenes infections from an unidentified source continues with the client count now at21 The FDA started the examination on Feb. 13 and has actually started traceback efforts however has actually not reported what is being traced. The company has actually not launched any other information about the scenario.
Abbott baby formula break out
The FDA continues to examine a break out of cronobacter infections connected to infant formula produced by Abbott Nutrition’s Sturgis, MI, plant. According to FDA edited files gotten by eFoodAlert there have actually been 9 crib death related to the examination.
The Abbott production plant, that makes Similac and Elecare solutions in addition to other brand names, was closed in mid-February after the FDA discovered 5 pressures of cronobacter in the center. At that point the FDA was just reporting 4 children connected with the break out with 2 having passed away.
FDA authorities accepted enable production at the plant to resume on June 4 with stringent controls in location.
Other active break out examinations
A break out of Salmonella infections traced to Jif peanut butter continues under examination by the FDA. It has actually sickened 16 individuals in 12 mentions since the most current report from the Centers for Disease Control and Prevention. Lots of Jif branded items have actually been remembered and lots of items including the linked Jif peanut butter have actually likewise been remembered.
An examination into 558 “negative occasion reports” about dry cereal, Lucky Charms breakfast cereal, is continuous. The FDA has actually started on-site of General Mills production plant and is carrying out ecological and item screening. No other info abut the break out of “unfavorable occasions” has actually been launched.
Additional break out details
The table listed below programs info about break out examinations being handled by FDA’s CORE Response Teams. The examinations remain in a range of phases. Some break outs have actually restricted info with active examinations continuous, others might be near conclusion. The table listed below has actually been abbreviated to reveal just active examinations.
A public health advisory will be released for examinations that have actually led to particular, actionable actions for customers to require to safeguard themselves, according to the FDA. Please direct your attention to those pages for the most approximately date details on the examination and for customer defense details.
Outbreak and negative occasion examinations that do not lead to particular, actionable actions for customers might or might not conclusively determine a source or expose any contributing elements. Unfavorable occasion examinations depend on self-reported information. These reports might call a specific item, FDA will just show an item classification in the table and will not openly call a particular item till there is adequate proof to link that item as a cause of health problems or negative occasions. If a cause and/or contributing aspects are recognized that might notify future avoidance, FDA devotes to supplying a summary of those findings.
To see the FDA page with links to particular details on specific break outs, please click on this link.
( To register for a complimentary membership to Food Safety News, click here)
Source: FDA examining brand-new break out of E. coli O157: H7 infections; 10 individuals ill